A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine is
AIEEE 2003, Medium
(b) $\eta = 1 - \frac{{{T_2}}}{{{T_1}}} = \frac{W}{Q}$
==>$W = \left( {1 - \frac{{{T_1}}}{{{T_2}}}} \right)\;Q = \left\{ {1 - \frac{{(273 + 27)}}{{(273 + 627)}}} \right\}$
==>$W = \left( {1 - \frac{{300}}{{900}}} \right) \times 3 \times {10^6}$
$ = 2 \times {10^6} \times 4.2\;J = 8.4 \times {10^6}J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
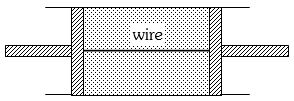
- 1A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
- 2View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 3How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
- 4$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
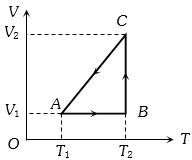
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 5A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 6An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 7A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
- 8Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
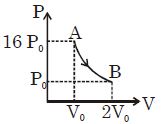
- 9Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
- 10An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
