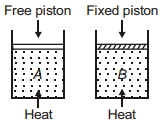
A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectively

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 2Find $V_B = ?$View Solution
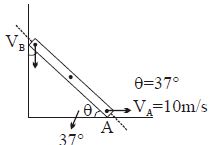
- 3A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
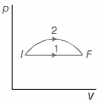
- 4An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution
- 5One gm mol of a diatomic gas $(\gamma = 1.4)$ is compressed adiabatically so that its temperature rises from ${27^o}C$ to ${127^o}C$. The work done will beView Solution
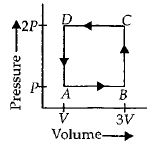
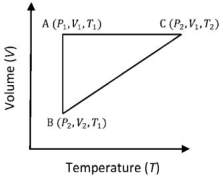
- 6A reversible cyclic process for an ideal gas is shown below. Here, $P , V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$ $(B)$ $W _{ BC }= P _2\left( V _2- V _1\right)$ and $q _{ BC }= H _{ AC }$ $(C)$ $\Delta H _{ CA }<\Delta U _{ CA }$ and $q _{ AC }=\Delta U _{ BC }$ $(D)$ $q_{B C}=\Delta H_{A C}$ and $\Delta H_{C A}>\Delta U_{C A}$
- 7View SolutionFirst law of thermnodynamics is given by
- 8View SolutionEntropy of a system decreases .........
- 9Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at ${T_1} = 800K$ and rejects to sink at ${T_2}K.$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at ${T_3} = 300K.$ If the work outputs of two engines are equal, then the value of ${T_2}$ is .... $K$View Solution
- 10Two cylinders contain same amount of ideal monatomic gas. Same amount of heat is given to two cylinders. If temperature rise in cylinder $A$ is $T_0$ then temperature rise in cylinder $B$ will be .........View Solution