A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$
Easy
(c)
$T_1=627+273=900\,K$
$Q_1=6000\,Cal$
$T_2=27+273=300\,K$
From Carnot Theorem $\frac{Q_1}{T_1}=\frac{Q_2}{T_2}$
$Q_2=\frac{T_2}{T_1} \times Q_1$
$=\frac{300}{900} \times 6000$
$=2000\,cal$
Work Done $=Q_1-Q_2$
$=6000-2000$
$=4000\,Cal$
$=4\,Kcal$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
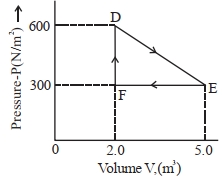
- 2A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution
- 3View SolutionIf an ideal gas is compressed isothermally then
- 4Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 5An ideal monoatomic gas expands to twice its volume. If the process is isothermal, the magnitude of work done by the gas is $W_i$. If the process is adiabatic, the magnitude of work done by the gas is $W_a$. Which of the following is true?View Solution
- 6View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
- 7Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
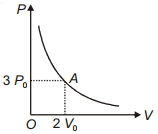
- 8The variation of pressure $P$ with volume $V$ for an ideal monatomic gas during an adiabatic process is shown in figure. At point $A$ the magnitude of rate of change of pressure with volume isView Solution
- 9View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 10View SolutionWhich of the following can be coefficient of performance of refrigerator?
