Match List $I$ with List $II$ :
| List $I$ | List $II$ |
| $A$ Isothermal Process | $I$ Work done by the gas decreases internal energy |
| $B$ Adiabatic Process | $II$ No change in internal energy |
| $C$ Isochoric Process | $III$ The heat absorbed goes partly to increase internal energy and partly to do work |
| $D$ Isobaric Process | $IV$ No work is done on or by the gas |
Choose the correct answer from the options given below :
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
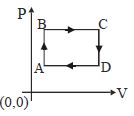
- 2The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
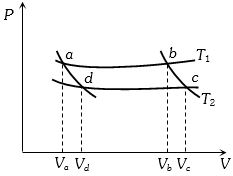
- 3In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
- 4A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 5View SolutionIn a thermodynamic system working substance is ideal gas, its internal energy is in the form of
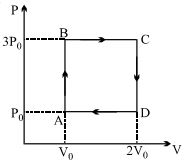
- 6A ideal monoatomic gas is carried around the cycle $ABCDA$ as shown in the fig. The efficiency of the gas cycle isView Solution
- 7An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
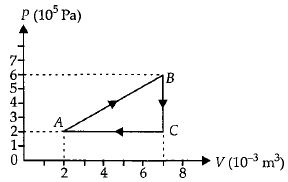
- 8A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 9View SolutionIn the following indicator diagram, the net amount of work done will be
- 10For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
