A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$
Medium
(c) $\eta = 1 - \frac{{{T_2}}}{{{T_1}}} = 1 - \frac{{(273 + 69)}}{{(273 + 411)}} = 0.5$
==> Work done$ = \eta \times Q = 0.5 \times 1000 = 500\,J$
==> Work done$ = \eta \times Q = 0.5 \times 1000 = 500\,J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
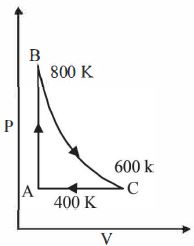
- 1One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statementView Solution
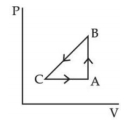
- 2A sample of an ideal gas is taken through the cyclic process $ABCA$ as shown in figure. It absorbs, $40\,J$ of heat during the part $A B$, no heat during $BC$ and rejects $60\,J$ of heat during $CA$. $A$ work $50\,J$ is done on the gas during the part $BC$. The internal energy of the gas at $A$ is $1560\,J$. The work done by the gas during the part $CA$ is.............$J$View Solution
- 3A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
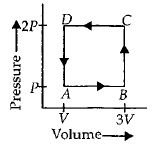
- 4A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle isView Solution
- 5View SolutionFor an ideal gas, in an isothermal process
- 6A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 7View SolutionWork done on or by a gas, in general depends upon the
- 8A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 9The specific heat of hydrogen gas at constant pressure is ${C_P} = 3.4 \times {10^3}cal/kg{\,^o}C$ and at constant volume is ${C_V} = 2.4 \times {10^3}cal/kg{\,^o}C.$If one kilogram hydrogen gas is heated from ${10^o}C$ to ${20^o}C$ at constant pressure, the external work done on the gas to maintain it at constant pressure isView Solution
- 10An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
