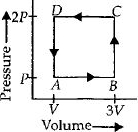
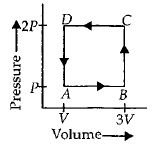
A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle is

AIPMT 2012, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 2In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
- 3$Assertion :$ Reversible systems are difficult to find in real world.View Solution
$Reason :$ Most processes are dissipative in nature. - 4An ideal gas with constant heat capacity $C_V=\frac{3}{2} n R$ is made to carry out a cycle that is depicted by a triangle in the figure given below. The following statement is true about the cycle.View Solution
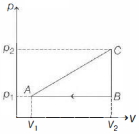
- 5View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
- 6A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
- 7If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
- 8An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
- 9A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine isView Solution
- 10The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution