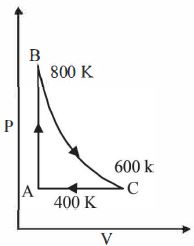
One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statement

JEE MAIN 2014, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 2If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, thenView Solution
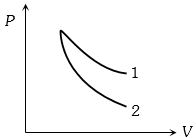
- 3$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
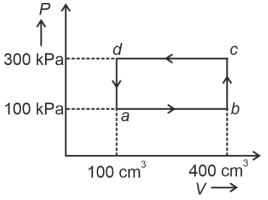
- 4A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ isView Solution
- 5View SolutionThe internal energy of the gas increases In
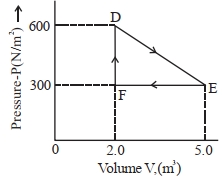
- 6A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution
- 7Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 8One mole of a gas expands obeying the relation as shown in the $P/V$ diagram. The maximum temperature in this process is equal toView Solution
- 9View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 10An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
