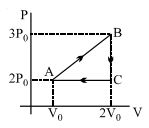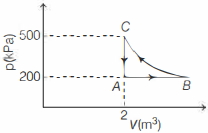
$\ln$ isothermal process $B C$,
$p V=\text { constant }$
$\Rightarrow \quad D_C V_C=p_B V_B$
$\Rightarrow \quad \frac{p_C V_C-500 \times 2}{p_B}=5\, m ^3$
Now, work done in the complete cycle $C A B C$ is
$W_{C A B C}=W_{C A}+W_{A B}+W_{B C}$
$=0+p \Delta V+\int p d V$
$=200(5-2)+\int \limits_5^2 k \frac{d V}{V}$
$=600+k \int \limits_5^2 \frac{d V}{V}$
$=600+1000(\log 2-\log 5)$
$=600+1000(0.69-160)$
$=600-910 \approx-300 \,kJ$
Download our appand get started for free
Similar Questions
- 1$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 2A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 3View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 4Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
- 5A refrigerator is to maintain eatables kept inside at $9^{\circ} C .$ If room temperature is $36^{\circ} C$ calculate the coefficient of performance.View Solution
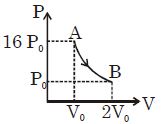
- 6Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution
- 7View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 8Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
- 9The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$View Solution
- 10View SolutionIn the above thermodynamic process, the correct statement is