The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionOut of the following which quantity does not depend on path
- 2View SolutionWhich of the following is not a thermodynamics co-ordinate
- 3View SolutionThe internal energy of the gas increases In
- 4A Carnot engine operates between two reservoirs of temperatures $900\; \mathrm{K}$ and $300 \;\mathrm{K}$ The engine performs $1200\; \mathrm{J}$ of work per cycle. The heat energy (in $\mathrm{J}$ ) delivered by the engine to the low temperature reservoir, in a cycle. isView Solution
- 5This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1:$ In an adiabatic process, change in internal energy of a gas is equal to work done on/by the gas in the process.Statement $2 :$ The temperature of a gas remains constant in an adiabatic process.
- 6View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
- 7View SolutionA gas undergoes a change at constant temperature. Which of the following quantities remain fixed?
- 8The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 9Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
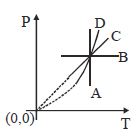
- 10Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
