A cylinder with fixed capacity of $67.2\, lit$ contains helium gas at $STP$. The amount of heat needed to raise the temperature of the gas by $20\,^oC$ is ..... $J$ [Given that $R = 8.31\, J\, mol^{-1}\, K^{-1}$]
JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The graph between pressure $(P)$ and $\frac{1}{V}$ ($V$ is volume) for constant temperature process in ideal equation $(PV = nRT)$View Solution
- 2The temperature at which root mean square speed of a gas will be half of its value at $0\,^oC$ is ....... $^oC$View Solution
- 3View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
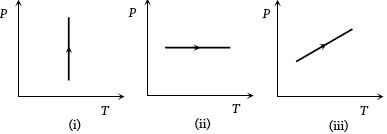
- 4If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
- 5$2\, moles$ of helium gas is mixed with $3\, moles$ of hydrogen molecules (taken to be rigid). What is the molar specific heat of mixture at constant volume ....... $J/mol\, K$ ? $(R = 8.3\, J/mol\, K)$View Solution
- 6At a certain temperature, the $r.m.s.$ velocity for ${O_2}$ is $400\, m/sec.$ At the same temperature, the $r.m.s.$ velocity for ${H_2}$ molecules will be ....... $m/sec$View Solution
- 7A container has $N$ molecules at absolute temperature $T$. If the number of molecules is doubled but kinetic energy in the box remain the same as before, the absolute temperature of the gas is ...........View Solution
- 8One mole of monatomic gas and three moles of diatomic gas are put together in a container. The molar specific heat (in $JK ^{-1} mol ^{-1}$ ) at constant volume is (Let $R=8 \,JK ^{-1} mol ^{-1}$ )View Solution
- 9View SolutionWhich of the following statements is/are not an assumption of the kinetic theory for an ideal gas?
- 10The volume of a gas at $20°C$ is $200\, ml$. If the temperature is reduced to $-20°C$ at constant pressure, its volume will be ...... $ml$View Solution
