At a certain temperature, the $r.m.s.$ velocity for ${O_2}$ is $400\, m/sec.$ At the same temperature, the $r.m.s.$ velocity for ${H_2}$ molecules will be ....... $m/sec$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of hydrogen and oxygen has volume $2000 \; cm ^{3}$, temperature $300 \; K$, pressure $100 \; kPa$ and mass $0.76 \; g$ The ratio of number of moles of hydrogen to number of moles of oxygen in the mixture will beView Solution
- 2Three closed vessels $A, B$ and $C$ are at the same temperature $T$ and contain gases which obey the Maxwellian distribution of velocities. Vessel $A$ contains only ${O_2},\,B$ only ${N_2}$ and $C$ a mixture of equal quantities of ${O_2}$ and ${N_2}$. If the average speed of the ${O_2}$ molecules in vessel A is ${V_1}$, that of the ${N_2}$ molecules in vessel B is ${V_2}$, the average speed of the ${O_2}$ molecules in vessel $C$ isView Solution
- 3Find the ratio of specific heat at constant pressure to the specific heat constant volume for $N{H_3}$View Solution
- 4View SolutionIf hydrogen gas is heated to a very high temperature, then the fraction of energy possessed by gas molecules correspond to rotational motion ...........
- 5A mixture of hydrogen and oxygen has volume $500\, {cm}^{3}$, temperature $300\, {K}$, pressure $400\, {kPa}$ and mass $0.76\, {g}$. The ratio of masses of oxygen to hydrogen will be :-View Solution
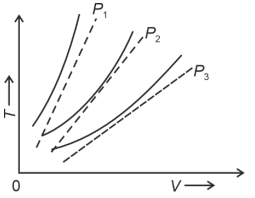
- 6The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 7If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
- 8A closed vessel contains $10 g$ of an ideal gas $X$ at $300 \ K$, which exerts $2$ atm pressure. At the same temperature, $80 g$ of another ideal gas $Y$ is added to it and the pressure becomes $6 atm$. The ratio of root mean square velocities of $X$ and $Y$ at $300 \ K$ isView Solution
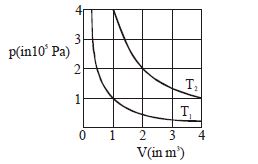
- 9The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution
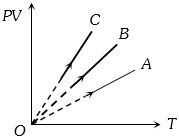
- 10$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution