A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:
JEE MAIN 2024, Diffcult
$\gamma=1+\frac{2}{f}=1.4 \Rightarrow \frac{2}{f}=0.4$
$\Rightarrow f=5$
$W=n R \Delta T=200 J$
$Q=\left(\frac{f+2}{2}\right) n R \Delta T$
$=\frac{7}{2} \times 200=700 J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 2Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
- 3When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
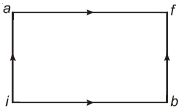
- 4A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
- 5$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 6Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 7Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 8For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
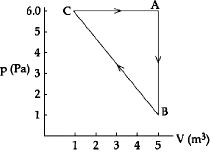
- 9$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 10The specific heat of hydrogen gas at constant pressure is ${C_P} = 3.4 \times {10^3}cal/kg{\,^o}C$ and at constant volume is ${C_V} = 2.4 \times {10^3}cal/kg{\,^o}C.$If one kilogram hydrogen gas is heated from ${10^o}C$ to ${20^o}C$ at constant pressure, the external work done on the gas to maintain it at constant pressure isView Solution
