$Assertion :$ The isothermal curves intersect each other at a certain point.
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope.
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope.
AIIMS 2001,AIIMS 2008, Easy
Isothermal curves have slope which is equal to $\frac{P}{V}$. It can be calculated as follows $PV = RT$ Differentiating, $PdV + VdP = 0$ $-\frac{dP}{dV} = \frac{P}{V}$ Now, if they cut each other at certain point, they will have different slope at the same point (for same value of $P$ and $V$). So, they can not cut each other at some point. Reason is true, slope is $\frac{P}{V}$ . For adiabatic curve slope is $\gamma $ times $\frac {P}{V}$ .
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe heat absorbed by a system in going through the given cyclic process is:
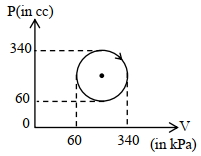
- 2Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
- 3A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
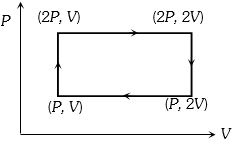
- 4Work done in the given $P-V$ diagram in the cyclic process isView Solution
- 5A bicycle tyre is filled with air having pressure of $270\,kPa$ at $27^{\circ}\,C$. The approximate pressure of the air in the tyre when the temperature increases to $36^{\circ}\,C$ is $............kPa$View Solution
- 6View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 7View SolutionFor which combination of working temperatures the efficiency of Carnot’s engine is highest
- 8Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 9An ideal gas is subjected to a thermodynamic process $PV^{2.5} = 0.40$ where $P$ is in $Pa$ and $V$ is in $m^3$.What is the slope of the $P-V$ curve with volume plotted against $x-$ axis at $V=1\, m^3$ ?View Solution
- 10View SolutionIn thermodynamic processes which of the following statements is not true?
