A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will be
AIPMT 1996, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
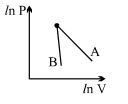
- 1The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 2If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 3$1\, mole$ of an ideal monoatomic gas at temperature $'T_0'$ expands slowly according to the law $P = KV$, where $K$ is a constant. If the final temperature of the gas is $2T_0$. Find the heat supplied to the gasView Solution
- 4A Carnot engine absorbs an amount $Q$ of heat from a reservoir at an absolute temperature $T$ and rejects heat to a sink at a temperature of $T/3$ . The amount of heat rejected isView Solution
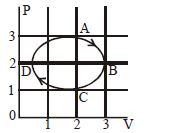
- 5The figure shows the $P-V$ plot of an ideal gas taken through a cycle $ABCDA.$ The part $ABC$ is a semicircle and $CDA$ is half of an ellipse. Then,View Solution
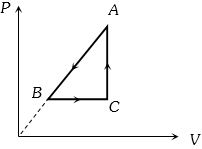
- 6$P-V$ diagram of a cyclic process $ABCA$ is as shown in figure. Choose the correct statementView Solution
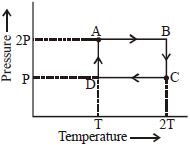
- 7An ideal monoatomic gas is taken through the thermodynamic states $A \to B \to C \to D$ via the paths shown in the figure. If $U_A, U_B, U_C$ and $U_D$ represent the internal energy of the gas in state $A, B\, C$ and $D$ respectively, then which of the following is not true?View Solution
- 8An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)View Solution
- 9For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 10The adiabatic elasticity of a diatomic gas at $NTP$ is ........ $N / m ^2$View Solution
