An ideal gas at a pressures of $1$ atmosphere and temperature of ${27^o}C$ is compressed adiabatically until its pressure becomes $8$ times the initial pressure, then the final temperature is ..... $^oC$ ($\gamma = 3/2$)
Medium
(d)Using relation$\frac{{{T_2}}}{{{T_1}}} = {\left( {\frac{{{P_2}}}{{{P_1}}}} \right)^{\frac{{\gamma - 1}}{\gamma }}} = {(8)^{\frac{{3/2 - 1}}{{3/2}}}} = 2$.
==> ${T_2} = 2{T_1}$==> ${T_2} = 2\,(273 + 27) = 600K = 327^\circ C$
==> ${T_2} = 2{T_1}$==> ${T_2} = 2\,(273 + 27) = 600K = 327^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 2The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
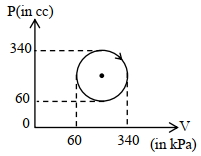
- 3View SolutionThe heat absorbed by a system in going through the given cyclic process is:
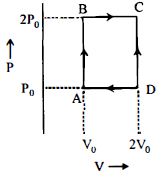
- 4An engine operates by taking $n\,moles$ of an ideal gas through the cycle $ABCDA$ shown in figure. The thermal efficiency of the engine is : (Take $C_v =1 .5\, R$, where $R$ is gas constant)View Solution
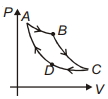
- 5Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 6A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 7An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
- 8View SolutionAn adiabatic process occurs at constant
- 9A gas is enclosed in a cylinder with a movable frictionless piston. Its initikl thermodynamic state at pressure $P_i=10^5 \mathrm{~Pa}$ and volume $V_i=10^{-3} \mathrm{~m}^3$ chanıes to i final state at $P_f=(1 / 32) \times 10^5 \mathrm{~Pa}$ and $V_f=8 \times 10^{-3} \mathrm{~m}^3$ in an adiabatic quasi-static process, such that $P^3 V^5=$ constant. Consider another thermodynamic process that brings the system from the same initial state to the same final state in two steps: an isobaric expansion at $P$, followed by an isochoric (isovolumetric) process at volume $V_f$. The amount of heat supplied to the system in the two-step process is approximatelyView Solution
- 10View SolutionIf a gas is heated at constant pressure, its isothermal compressibility
