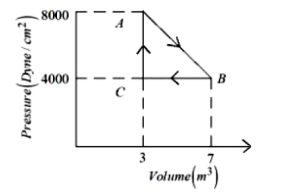
A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$

Medium
$\mathrm{AB} \rightarrow \Delta \mathrm{Q}=\mu \mathrm{C}_{\mathrm{v}} \Delta \mathrm{T}$
$=\mu \frac{1}{2} \mathrm{R}\left(2 \mathrm{T}_{0}-\mathrm{T}_{0}\right)$
$=\frac{5}{2} \mathrm{P}_{0} \mathrm{V}_{0}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
- 2An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution
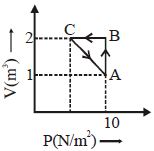
- 3The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
- 4An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
- 5The value of $\eta$ may lie betweenView Solution
- 6The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
- 7View SolutionSelect the correct statement for work, heat and change in internal energy.
- 8View SolutionWhich statement is incorrect?
- 9Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 10A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution