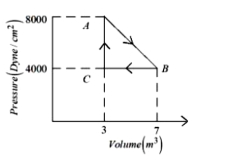
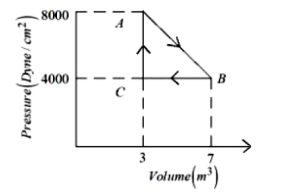
A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :

JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
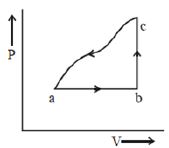
- 2A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 3This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
- 4Heat is given to an ideal gas in an isothermal process.View Solution
$A.$ Internal energy of the gas will decrease.
$B.$ Internal energy of the gas will increase.
$C.$ Internal energy of the gas will not change.
$D.$ The gas will do positive work.
$E.$ The gas will do negative work.
Choose the correct answer from the options given below :
- 5View SolutionFor adiabatic process, wrong statement is
- 6$Assertion :$ The isothermal curves intersect each other at a certain point.View Solution
$Reason :$ The isothermal change takes place slowly, so, the isothermal curves have very little slope. - 7View SolutionAn ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
- 8The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
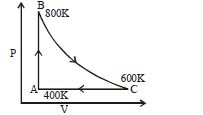
- 9One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 10View SolutionWhich of the following is not thermodynamical function