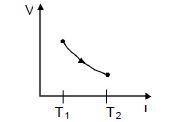
The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)

Diffcult
$\mathrm{VT}=\mathrm{C}_{0}$
$\mathrm{PV}^{2}=\mathrm{C}_{0}, \quad \mathrm{x}=2$
$C=C_{v}+\frac{R}{1-r}=\left(\frac{3 R}{2}\right)$
$W=\left[\frac{n R \Delta T}{(-x)}\right]=\frac{2 R \times 200}{(1-2)}=-400 R$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Three samples of the same gas $A, B$ and $C(\gamma = 3/2)$ have initially equal volume. Now the volume of each sample is doubled. The process is adiabatic for $A$ isobaric for $B $ and isothermal for $C$. If the final pressures are equal for all three samples, the ratio of their initial pressures areView Solution
- 2A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
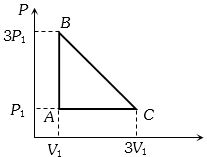
- 3An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
- 4A scientist says that the efficiency of his heat engine which operates at source temperature $127°C$ and sink temperature $27°C$ is $26\%$, thenView Solution
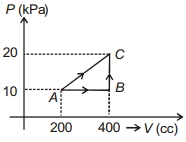
- 5If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
- 6A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )View Solution
- 7Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

- 8The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
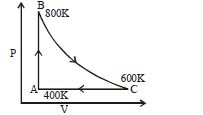
- 9One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 10A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$View Solution
