A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
- 2One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$. The change in the internal energy of the gas is .... $J$ (given $R = 8.3 \,J/mole/K$)View Solution
- 3View SolutionIf a system undergoes contraction of volume then the work done by the system will be
- 4A constant amount of an ideal gas undergoes the cyclic process $A B C A$ in the $p-V$ graph shown below. The path $B C$ is an isothermal. The work done by the gas during one complete cycle, beginning and ending at $A$ is nearly .......... $\,kJ$View Solution
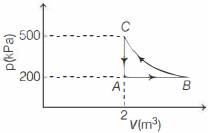
- 5Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
- 6View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 7$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
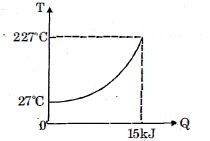
- 8During the adiabatic expansion of $2$ moles of a gas, the internal energy of the gas is found to decrease by $2$ joules, the work done during the process on the gas will be equal to ....... $J$View Solution
- 9A sample of an ideal gas is taken through the cyclic process $ABCA$ as shown in figure. It absorbs, $40\,J$ of heat during the part $A B$, no heat during $BC$ and rejects $60\,J$ of heat during $CA$. $A$ work $50\,J$ is done on the gas during the part $BC$. The internal energy of the gas at $A$ is $1560\,J$. The work done by the gas during the part $CA$ is.............$J$View Solution
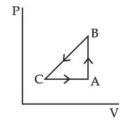
- 10View SolutionA gas is being compressed adiabatically. The specific heat of the gas during compression is
