This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
AIEEE 2012, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
- 2View SolutionThe first operation involved in a Carnot cycle is
- 3An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1\,g\,{s^{ - 1}}$. The calorific value of fuel is $2k cal/g$. His claimView Solution
- 4In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
- 5The efficiency of carnot engine is $50\%$ and temperature of sink is $500\,K$ . If temperature of source is kept constant and its efficiency raised to $60\%$ , then the required temperature of the sink will be .... $K$View Solution
- 6A Carnot’s engine is made to work between $200°C$ and $0°C$ first and then between $0°C$ and $-200°C.$ The ratio of efficiencies of the engine in the two cases isView Solution
- 7View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
- 8A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 9View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
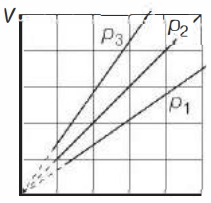
- 10View SolutionWhich of the following statements is correct for any thermodynamic system
