A gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then
NEET 2016, Easy
${V_1} = V,{V_2} = V/2$
$On\,P - V\,diagram,$
$Area\,under\,adiabatic\,curve>Area\,under\,isothermal\,curve,$
So compressing the gas through adiabatic process will require more work to be done.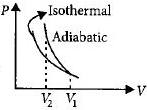
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIf an ideal gas is compressed isothermally. Which of the following statements is true?
- 2An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 3The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 4A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 5View SolutionThe internal energy of the gas increases In
- 6A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 7A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
- 8Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 9View SolutionThe specific heat of a gas in an isothermal process is
- 10A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
