If an ideal gas is compressed isothermally. Which of the following statements is true?
KVPY 2021, Easy
(D)
$Q =\Delta U + W$
For isothermal process $\Delta U =0$
$\therefore$ Internal energy remains constant
$Q = W$
Positive work done by external agent is completely lost in the form of heat.
As volume gets reduced, pressure increases
$( PV =$ constant $)$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 2An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 3The initial pressure and volume of an ideal gas are $P_0$ and $V_0$. The final pressure of the gas when the gas is suddenly compressed to volume $\frac{ V _0}{4}$ will be (Given $\gamma=$ ratio of specific heats at constant pressure and at constant volume)View Solution
- 4If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
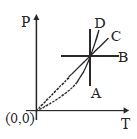
- 5View SolutionPressure versus temperature graph of an ideal gas is shown in figure
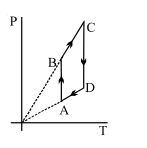
- 6Carnot heat engine works with an ideal diatomic gas and an adiabatic volume expansion ratio $32$ . Then its efficiency is ....... $\%$View Solution
- 7View SolutionThe rate of recombination or generation are governed by the law(s) of
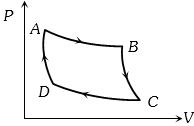
- 8Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
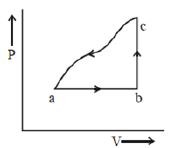
- 9A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 10Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution