Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
- 2View SolutionA gas is compressed adiabatically, which one of the following statement is NOT true.
- 3One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
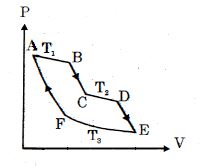
- 4One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas
- 5View SolutionCompressed air in the tube of a wheel of a cycle at normal temperature suddenly starts coming out from a puncture. The air inside
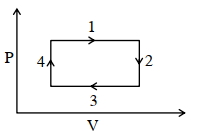
- 6An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
- 7In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 8A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
- 9A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 10The temperature of sink of Carnot engine is ${27^o}C$. Efficiency of engine is $25\%.$ Then temperature of source is ...... $^oC$View Solution
