Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
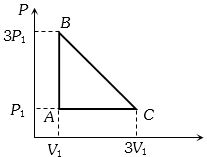
- 1An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
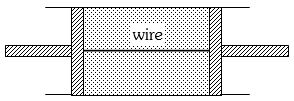
- 2A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
- 3If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$View Solution
- 4During an experiment, an ideal gas is found to obey a condition $VP^2 =$ constant. The gas is initially at a temperature $T$, pressure $P$ and volume $V.$ The gas expands to volume $4V$.View Solution
- 5An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
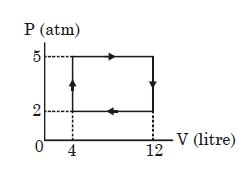
- 6View SolutionAn ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
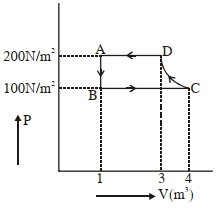
- 7The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$View Solution
- 8A solid body of constant heat capacity $1\,J /{ }^{\circ} C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with 8 reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^{\circ}\,C$ to final temperature $200^{\circ}\,C$. Entropy change of the body in the two cases respectively is :
- 9An engine operating between the boiling and freezing points of water will haveView Solution
$1.$ efficiency more than $27 \%$
$2.$ efficiency less than the efficiency a Carnot engine operating between the same two temperatures.
$3.$ efficiency equal to $27 \%$
$4.$ efficiency less than $27 \%$
- 10The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution