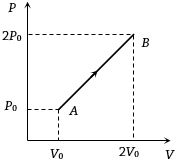
The $P-V$ diagram of $2$ gm of helium gas for a certain process $A \to B$ is shown in the figure. what is the heat given to the gas during the process $A \to B$

Medium
(b) Change in internal energy from $A \to B$ is
$\Delta U = \frac{f}{2}\mu R\Delta T = \frac{f}{2}({P_f}{V_f} - {P_i}{V_i})$
$ = \frac{3}{2}(2{P_0} \times 2{V_0} - {P_0} \times {V_0}) = \frac{9}{2}{P_0}{V_0}$
Work done in process $A \to B$ is equal to the Area covered by the graph with volume axis i.e.,
${W_{A \to B}} = \frac{1}{2}({P_0} + 2{P_0}) \times (2{V_0} - {V_0}) = \frac{3}{2}{P_0}{V_0}$
Hence, $\Delta Q = \Delta U + \Delta W$$ = \frac{9}{2}{P_0}{V_0} + \frac{3}{2}{P_0}{V_0} = 6{P_0}{V_0}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 2During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 3A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$View Solution
- 4Two identical containers $A$ and $B$ with frictionless pistons contain the same ideal gas at the same temperature and the same volume $V$. The mass of the gas in $A$ is ${m_A}$ and that in $B$ is ${m_B}$. The gas in each cylinder is now allowed to expand isothermally to the same final volume $2V$. The changes in the pressure in $A$ and $B$ are found to be $\Delta P$ and $1.5 \Delta P$ respectively. ThenView Solution
- 5Agas expands such that its initial and final temperature are equal. Also, the process followed by the gas traces a straight line on the $P-V$ diagram :View Solution
- 6An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 7Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
- 8Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 9If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
- 10An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
