If the temperature of sink is at absolute zero, then the efficiency of Carnot engine will be ........ $\%$
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
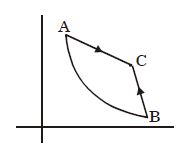
- 2A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
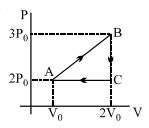
- 3A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 4View SolutionThe first operation involved in a Carnot cycle is
- 5Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
- 6A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
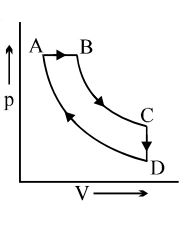
- 7The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
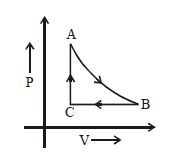
- 8An ideal gas expands isothermally from a volume ${V_1}$ to ${V_2}$ and then compressed to original volume ${V_1}$adiabatically. Initial pressure is ${P_1}$ and final pressure is ${P_3}$. The total work done is $W$. ThenView Solution
- 9For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 10View SolutionIn the above thermodynamic process, the correct statement is