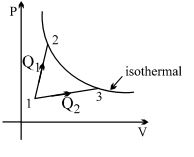
A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. Then

Advanced
Let the volumes and temperatures of gas at points 1,2 and 3 are $V_1, T_1, V_2, T_2$ and $V _3, T _3$ respectively.
Given: $T_2=T_3$
Thus $\Delta U _1= nC _{ v }\left( T _2- T _1\right)$
Also $\Delta U _2= nC _{ v }\left( T _3- T _1\right)$
But $T _2= T _3 \Rightarrow \Delta U _1=\Delta U _2$
Work done by the gas in process 1, $W _1= nRT \ln \frac{ V _2}{ V _1}$
Also work done by gas in process $2, \quad W_2=n R T \ln \frac{V_3}{V_1}$
$V _3 > V _2 \Rightarrow W _2 > W _1$
From ist law: $\quad Q=\Delta U+W$
$\therefore \quad Q _{ I }=\Delta U _1+ W _1$
Also $\quad Q _2=\Delta U _2+ W _2$
$\therefore Q _2- Q _1= W _2- W _1>0 \quad\left(\because \Delta U _1=\Delta U _2\right)$
$\Rightarrow Q _2 > Q _1$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 2The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 3An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$View Solution
- 4An ideal gas is taken through the cycle $A → B → C → A$, as shown in the figure. If the net heat supplied to the gas in the cycle is $5 \ J$, the work done by the gas in the process $C → A$ is ....... $J$View Solution
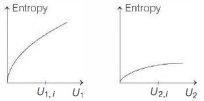
- 5Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 6Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
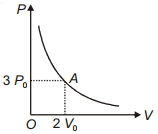
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 7The variation of pressure $P$ with volume $V$ for an ideal monatomic gas during an adiabatic process is shown in figure. At point $A$ the magnitude of rate of change of pressure with volume isView Solution
- 8Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 9One mole of an ideal gas $(\gamma = 1.4)$ is adiabatically compressed so that its temperature rises from $27\,^oC$ to $35\,^oC$ . The change in the internal energy of the gas is .... $J$. (given $R = 8.3\,J/mole-K$ )View Solution
- 10View SolutionWhich is incorrect
