Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
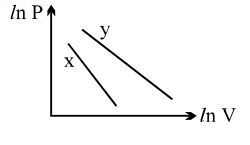
- 1For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
- 2A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 3View SolutionA process can be reversible if
- 4$1\, mole$ of an ideal monoatomic gas at temperature $'T_0'$ expands slowly according to the law $P = KV$, where $K$ is a constant. If the final temperature of the gas is $2T_0$. Find the heat supplied to the gasView Solution
- 5View SolutionWhich of the following is not a thermodynamics co-ordinate
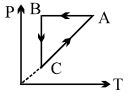
- 6A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 7View SolutionA container that suits the occurrence of an isothermal process should be made of
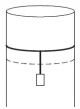
- 8A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 9View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
- 10$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
