An ideal heat engine exhausting heat at ${77^o}C$ is to have a $30\%$ efficiency. It must take heat at ...... $^oC$
Medium
(b) $\eta = 1 - \frac{{{T_2}}}{{{T_1}}}$ ==> $\frac{{30}}{{100}} = 1 - \frac{{350}}{{{T_1}}}$
==> $\frac{{350}}{{{T_1}}} = 1 - \frac{{50}}{{100}} = \frac{{70}}{{100}} = \frac{7}{{10}}$==> ${T_1} = 500\,K = 227^\circ C$
==> $\frac{{350}}{{{T_1}}} = 1 - \frac{{50}}{{100}} = \frac{{70}}{{100}} = \frac{7}{{10}}$==> ${T_1} = 500\,K = 227^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
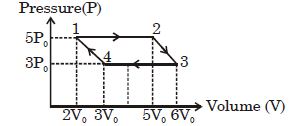
- 2An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
- 3Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
- 4View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
- 5View SolutionThe specific heat of a gas in an isothermal process is
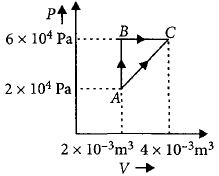
- 6Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
- 7When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 8Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
- 9A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process? $( J =4.18$ $Joules / cal )$ (in joule)View Solution
- 10Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
