A given ideal gas with $\gamma = \frac{{{C_p}}}{{{C_v}}} = 1.5$ at a temperature $T$. If the gas is compressed adiabatically to one-fourth of its initial volume, the final temperature will be ..... $T$
AIEEE 2012, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
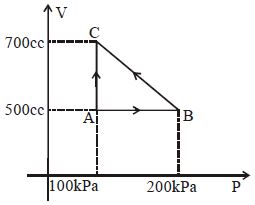
- 2An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
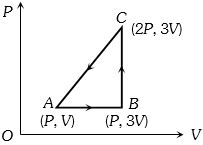
- 3View SolutionIrreversible process is
- 4A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
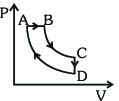
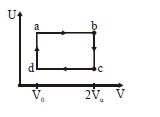
- 5The figure given below shows the variation in the internal energy $U$ with volume $V$ of $2.0\ mole$ of an ideal gas in a cyclic process $abcda$ . The temperatures of the gas during the processes $ab$ and $cd$ are $500\ K$ and $300\ K$ respectively, the heat absorbed by the gas during the complete process is .... $J$View Solution
(Take $R$ = $8.3\ J/mol-K$ and $ln\ 2$ = $0.69$)
- 6A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :View Solution
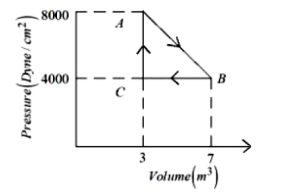
- 7In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $30$ joules of heat and $10$ joules of work was done on the gas. If the initial internal energy of the gas was $30$ joules, then the final internal energy will be ........ $J$View Solution
- 8A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 9The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
- 10The temperature inside and outside a refrigerator are $273 \,K$ and $300 \,K$ respectively. Assuming that the refrigerator cycle is reversible, for every joule of work done, the heat delivered to the surrounding will be nearly ........ $J$View Solution
