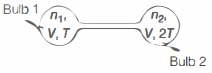
Two bulbs of identical volumes connected by a small capillary are initially filled with an ideal gas at temperature $T$. Bulb $2$ is heated to maintain a temperature $2 T$, while bulb $1$ remains at temperature $T$. Assume throughout that the heat conduction by the capillary is negligible. Then, the ratio of final mass of the gas in bulb $2$ to the initial mass of the gas in the same bulb is close to
KVPY 2011, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA diatomic molecule has how many degrees of freedom
- 2A gas has $n$ degrees of freedom. The ratio of specific heat of gas at constant volume to the specific heat of gas at constant pressure will be.View Solution
- 3When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 4View SolutionThe time average of the kinetic energy of one molecule of a gas taken over a long period of time
- 5The temperature at which the average translational kinetic energy of a molecule is equal to the energy gained by an electron in accelerating from rest through a potential difference of $1\, volt$ isView Solution
- 6The total kinetic energy of translatory motion of all the molecules of $5$ litres of nitrogen exerting a pressure $P$ is $3000 \,\,J$.View Solution
- 7View SolutionThe pressure exerted by the gas on the walls of the container because
- 8A gas at $27^o C$ has a volume $V$ and pressure $P.$ On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ...... $^oC$View Solution
- 9The ratio of two specific heats $\frac{{{C_P}}}{{{C_V}}}$ of $CO$ isView Solution
- 10In a thermally isolated system, two boxes filled with an ideal gas are connected by a valve. When the valve is in closed position, states of the box $1$ and $2$ respectively, are ( $1 \,atm , V, T)$ and $(0.5 \,atm , 4 V, T)$. When the valve is opened, then the final pressure of the system is approximately ............... $atm$View Solution