For a gas, the $r.m.s.$ speed at $800\, K$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At a pressure of $24 \times {10^5}\,dyne/c{m^2}$, the volume of ${O_2}$ is $10\, litre$ and mass is $20\, gm.$ The $r.m.s.$ velocity will be....... $m/sec$View Solution
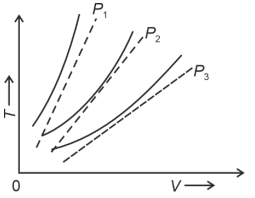
- 2The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 3An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
- 4The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at $0°C$ is $3180$ metres/second. The pressure on the hydrogen gas is ..... $atm$ (Density of hydrogen gas is $8.99 \times {10^{ - 2}}\,kg/{m^3}$, $1$ atmosphere $ = 1.01 \times {10^5}\,N/{m^2})$View Solution
- 5The temperature of the hydrogen at which the average speed of its molecules is equal to that of oxygen molecules at a temperature of $31\,^oC,$ is ........ $^oC$View Solution
- 6$Assertion :$ The ratio of $\frac{C_p}{C_v}$ for an ideal diatomic gas is less than that for an ideal monoatomic gas (where $C_p$ and $C_v$ have usual meaning).View Solution
$Reason :$ The atoms of a monoatomic gas have less degrees of freedom as compared to molecules of the diatomic gas. - 7The molecules of a given mass of a gas have a $r.m.s.$ velocity of $200\, m/sec$ at $27°C$ and $1.0 \times {10^5}\,N/{m^2}$ pressure. When the temperature is $127°C$ and pressure is $0.5 \times {10^5}\,N/{m^2}$, the $r.m.s.$ velocity in $m/sec$ will beView Solution
- 8The respective speeds of five molecules are $2, 1.5, 1.6, 1.6$ and $1.2 \,km/sec.$ The most probable speed in $km/sec$ will beView Solution
- 9$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:View Solution
- 10If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
