A litre of dry air at $STP$ expands adiabatically to a volume of $3$ litres. If $\gamma=1.40,$ the work done by air is$(3^{1.4}=4.6555)$ [Take air to be an ideal gas $]$
JEE MAIN 2020, Medium
$\mathrm{W}=\frac{\mathrm{nR}\left(\mathrm{T}_{1}-\mathrm{T}_{2}\right)}{\gamma-1}=\frac{\mathrm{P}_{1} \mathrm{V}_{1}-\mathrm{P}_{2} \mathrm{V}_{2}}{0.4}$
$W=\frac{100-\frac{100}{4.6555} \times 3}{0.4}=88.90\;J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn adiabatic expansion of a gas
- 2An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 3Even Carnot engine cannot give $100\%$ efficiency because we cannotView Solution
- 4Three samples of the same gas $A, B$ and $C(\gamma = 3/2)$ have initially equal volume. Now the volume of each sample is doubled. The process is adiabatic for $A$ isobaric for $B $ and isothermal for $C$. If the final pressures are equal for all three samples, the ratio of their initial pressures areView Solution
- 5View SolutionThe state of a thermodynamic system is represented by
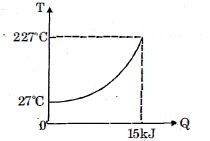
- 6$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
- 7One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 8View SolutionWhich of the following statements is correct for any thermodynamic system
- 9$Assertion :$ Reversible systems are difficult to find in real world.View Solution
$Reason :$ Most processes are dissipative in nature. - 10The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
