An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume is
Diffcult
$P T=$ constant
From ideal gas equation
$P V=n R T$
$T=\frac{P V}{n R}$
Put value of temperature in above equation. $P T=$ constant $P \frac{P V}{n R}=$ constant
$P^{2}=\frac{\text { constant }}{V}$
$P \propto \frac{1}{\sqrt{V}}$
Hence, the graph will be rectangular hyperbola.
Thus option $\mathrm{C}$ is correct.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 2If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 3$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 4View SolutionPressure versus temperature graph of an ideal gas is shown in figure
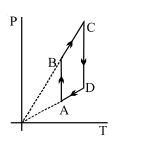
- 5A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
- 6An ideal gas goes through a reversible cycle $a\to b\to c\to d$ has the $V - T$ diagram shown below. Process $d\to a$ and $b\to c$ are adiabatic.... The corresponding $P - V$ diagram for the process is (all figures are schematic and not drawn to scale)View Solution
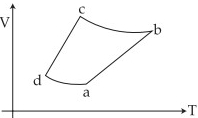
- 7View SolutionThe state of a thermodynamic system is represented by
- 8Consider the efficiency of Carnot's engine is given by $\eta=\frac{\alpha \beta}{\sin \theta} \log _{e} \frac{\beta x}{k T}$, where $\alpha$ and $\beta$ are constants. If $T$ is temperature, $k$ is Boltzman constant, $\theta$ is angular displacement and $x$ has the dimensions of length. Then, choose the incorrect option.View Solution
- 9In a thermodynamic process two moles of a monatomic ideal gas obeys $P \propto V^{-2}$. If temperature of the gas increases from $300 \,K$ to $400 \,K$, then find work done by the gas .........$R$ (where $R=$ universal gas constant).View Solution
- 10View SolutionTemperature is a measurement of coldness or hotness of an object. This definition is based on
