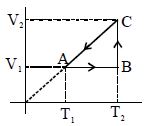
The state of a thermodynamic system is represented by
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 2Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
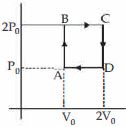
- 3Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at ${T_1} = 800K$ and rejects to sink at ${T_2}K.$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at ${T_3} = 300K.$ If the work outputs of two engines are equal, then the value of ${T_2}$ is .... $K$View Solution
- 4An ideal gas is taken through the cycle $A → B → C → A$, as shown in the figure. If the net heat supplied to the gas in the cycle is $5 \ J$, the work done by the gas in the process $C → A$ is ....... $J$View Solution
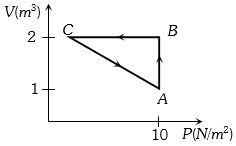
- 5Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 6View SolutionWhich of the following processes is reversible
- 7An ideal gas is made to undergo the cyclic process shown in the figure below. Let $\Delta W$ depict the work done, $\Delta U$ be the change in internal energy of the gas and $Q$ be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be (0 refers to no change)View Solution
- 8The volume of an ideal gas is $1$ litre and its pressure is equal to $72cm$ of mercury column. The volume of gas is made $900\, cm^3$ by compressing it isothermally. The stress of the gas will be ...... $cm$ (mercury)View Solution
- 9View SolutionThe internal energy of the gas increases In
- 10A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution