A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The rational between temperature and volume for the process is $TV^x =$ constant, then $x$ is
JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
- 2An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
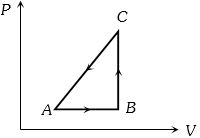
- 3The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
- 4One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 5A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
- 6A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$View Solution
- 7$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
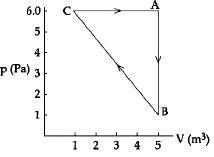
- 8For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
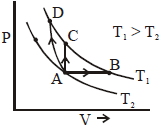
- 9Three different processes that can occur in an ideal monoatomic gas are shown in the $P$ vs $V$ diagram. The paths are labelled as $A \rightarrow B, A \rightarrow C$ and $A \rightarrow D .$ The change in internal energies during these process are taken as $E _{ AB }, E _{ AC }$ and $E _{ AD }$ and the work done as $W _{ AB }$ $W _{ AC }$ and $W _{ AD }$View Solution
The correct relation between these parameters are
- 10The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.View Solution
