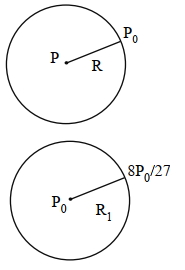
A spherical soap bubble inside an air chamber at pressure $P_0=10^5 Pa$ has a certain radius so that the excess pressure inside the bubble is $\Delta P=144 Pa$. Now, the chamber pressure is reduced to $8 P_0 / 27$ so that the bubble radius and its excess pressure change. In this process, all the temperatures remain unchanged. Assume air to be an ideal gas and the excess pressure $\Delta P$ in both the cases to be much smaller than the chamber pressure. The new excess pressure $\Delta P$ in $Pa$ is
IIT 2024, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of hydrogen and oxygen has volume $500\, {cm}^{3}$, temperature $300\, {K}$, pressure $400\, {kPa}$ and mass $0.76\, {g}$. The ratio of masses of oxygen to hydrogen will be :-View Solution
- 2The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
- 3Inside a cylinder having insulating walls and closed at ends is a movable piston, which divides the cylinder into two compartments. On one side of the piston is a mass $m$ of a gas and on the other side a mass $2 m$ of the same gas. What fraction of volume of the cylinder will be occupied by the larger mass of the gas when the piston is in equilibrium $?$ Consider that the movable piston is conducting so that the temperature is the same throughoutView Solution
- 4The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
- 5View SolutionWhich law states that effect of pressure is same for all portion
- 6A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 7A given mass of a gas is allowed to expand freely until its volume becomes double. If ${C_b}$ and ${C_a}$ are the velocities of sound in this gas before and after expansion respectively, then ${C_a}$ is equal toView Solution
- 8The average degree of freedom per molecule of a gas is $6$. The gas performs $25\ J$ work, while expanding at constant pressure. The heat absorbed by the gas is .... $J$View Solution
- 9View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
- 10At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution