A given mass of a gas is allowed to expand freely until its volume becomes double. If ${C_b}$ and ${C_a}$ are the velocities of sound in this gas before and after expansion respectively, then ${C_a}$ is equal to
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of gas is at $0°C.$ To what temperature it must be raised in order to double the $r.m.s.$ speed of the molecule ....... $^oC$View Solution
- 2When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution
- 3One mole of an ideal monoatomic gas at temperature $T_0$ expands slowly according to the law $P/V$ = constant. If the final temperature is $2 \,\,T_0$, heat supplied to the gas is :View Solution
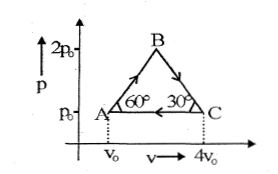
- 4Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 5If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
- 6Vessel $A$ is filled with hydrogen while vessel $B,$ whose volume is twice that of $A,$ is filled with the same mass of oxygen at the same temperature. The ratio of the mean kinetic energies of hydrogen and oxygen isView Solution
- 7At room temperature a diatomic gas is found to have an $r.m.s.$ speed of $1930\,ms^{-1}$. The gas isView Solution
- 8$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 9Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ isView Solution
- 10The temperature of a gas is doubled $(i)$ on absolute scale $(ii)$ on centigrade scale. The increase in root mean square velocity of gas will beView Solution
