A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is
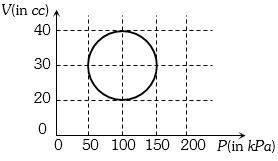

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A mixture of gases at $STP$ for which $\gamma=1.5$ is suddenly compressed to $\frac{1}{9}$ th of its original volume. The final temperature of mixture is .......... $^{\circ} C$View Solution
- 2A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
- 3One mole of an ideal gas at temperature $T_1$ expends according to the law $\frac{P}{{{V^2}}} =a$ (constant). The work done by the gas till temperature of gas becomes $T_2 $ isView Solution
- 4A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. ThenView Solution
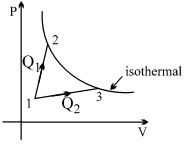
- 5The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
- 6Two samples $A$ and $B$ of a gas initially at the same pressure and temperature are compressed from volume $ V$ to $ V/2$ ($A$ isothermally and adiabatically). The final pressure of $ A$ isView Solution
- 7View SolutionA gas is being compressed adiabatically. The specific heat of the gas during compression is
- 8A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 9In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 10A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
