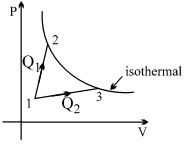
A gas takes part in two processes in which it is heated from the same initial state $1$ to the same final temperature. The processes are shown on the $P-V$ diagram by the straight line $1-2$ and $1-3$. $2$ and $3$ are the points on the same isothermal curve. $Q_1$ and $Q_2$ are the heat transfer along the two processes. Then

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionCofficient of performance of refigerator is
- 2For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
- 3An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 4A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 5$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):View Solution
- 6$300 \,cal$. of heat is given to a heat engine and it rejects $225 \,cal$. of heat. If source temperature is $227^{\circ} C$, then the temperature of sink will be____${ }^{\circ} C$.View Solution
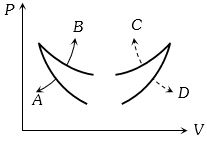
- 7Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
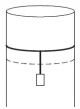
- 8A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 9View SolutionA Camot cycle consists of
- 10$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
