A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system is
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermally insulated rigid container contains an ideal gas heated by a filament of resistance $100 \,\Omega$ through a current of $1\,A$ for $5$ min then change in internal energy is...... $kJ$View Solution
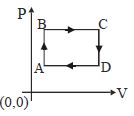
- 2The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
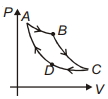
- 3Carnot cycle is plotted in $P-V$ graph. Which portion represents an isothermal expansion?View Solution
- 4View SolutionIn an isobaric expansion of an ideal gas, which of the following is zero?
- 5$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 6View SolutionWhen a gas expands adiabatically
- 7In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
- 8View SolutionSelect the correct statement for work, heat and change in internal energy.
- 9A mass of diatomic gas $(\gamma = 1 .4)$ at a pressure of $2$ atmospheres is compressed adiabatically so that its temperature rises from $27^o C$ to $927^o C.$ The pressure of the gas in the final state is ...... $atm$View Solution
- 10A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
