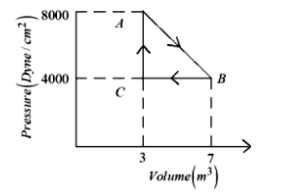
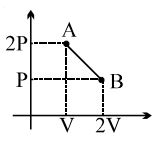
A thermodynamic system is taken from an original state $A$ to an intermediate state $B$ by a linear process as shown in the figure. It's volume is then reduced to the original value from $B$ to $C$ by an isobaric process. The total work done by the gas from $A$ to $B$ and $B$ to $C$ would be :

JEE MAIN 2024, Diffcult
Work done $\mathrm{AB}=\frac{1}{2}(8000+6000)$ Dyne $/ \mathrm{cm}^2 \times$ $4 \mathrm{~m}^3=\left(6000 \mathrm{Dyne} / \mathrm{cm}^2\right) \times 4 \mathrm{~m}^3$
Work done $\mathrm{BC}=-\left(4000\right.$ Dyne $\left./ \mathrm{cm}^2\right) \times 4 \mathrm{~m}^3$
Total work done $=2000$ Dyne $/ \mathrm{cm}^2 \times 4 \mathrm{~m}^3$
$=2 \times 10^3 $ $\times \frac{1}{10^5} \frac{\mathrm{N}}{\mathrm{cm}^2} \times 4 \mathrm{~m}^3 $
$ =2 \times 10^{-2} \times \frac{\mathrm{N}}{10^{-4} \mathrm{~m}^2} \times 4 \mathrm{~m}^3 $
$ =2 \times 10^2 \times 4 \mathrm{Nm}=800 \mathrm{~J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
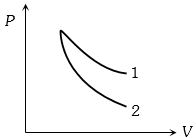
- 1$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
- 2If $300 ml$ of a gas at ${27^o}C$ is cooled to ${7^o}C$ at constant pressure, then its final volume will be ...... $ml$View Solution
- 3At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$View Solution
- 4The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 5$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
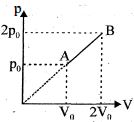
- 6An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
- 7$540$ calories of heat converts $1$ cubic centimeter of water at $100\,^oC$ into $1671$ cubic centimeter of steam at $100\,^oC$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly .......... $cal$View Solution
- 8An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 9The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 10An ideal Carnot engine, whose efficiency is $40 \%$ receives heat at $500\; K$. If its efficiency is $50 \%$ then the intake temperature for the same exhaust temperature is ......... $K$View Solution
