At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 2During an experiment, an ideal gas is found to obey a condition $VP^2 =$ constant. The gas is initially at a temperature $T$, pressure $P$ and volume $V.$ The gas expands to volume $4V$.View Solution
- 3An ideal gas at pressure $P$ and volume $V$ is expanded to volume$ 2V.$ Column $I$ represents the thermodynamic processes used during expansion. Column $II$ represents the work during these processes in the random order.:View Solution
Column $I$ Column $II$ $(p)$ isobaric $(x)$ $\frac{{PV(1 - {2^{1 - \gamma }})}}{{\gamma - 1}}$ $(q)$ isothermal $(y)$ $PV$ $(r)$ adiabatic (z) $PV\,\iota n\,2$ The correct matching of column $I$ and column $II$ is given by
- 4View SolutionThe specific heat of a gas in an isothermal process is
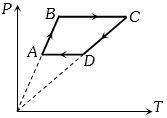
- 5Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution
- 6A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$View Solution
- 7Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 8$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 9View SolutionWhich of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
- 10A gas undergoes a change of state during which $100 J$ of heat is supplied to it and it does $20 J$ of work. The system is brought back to its original state through a process during which $20 J$ of heat is released by the gas. The work done by the gas in the second process is ....... $J$View Solution
