An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process is
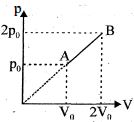

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas goes from state $A$ to state $B$ via three different processes as indicated in the $P-V$ diagram. If $Q_1, Q_2, Q_3$ indicate the heat absorbed by the gas along the three processes and $\Delta U_1, \Delta U_2, \Delta U_3$View Solution
indicate the change in internal energy along the three processes respectively, then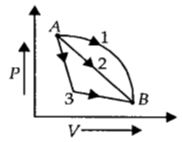
- 2A source supplies heat to a system at the rate of $1000 \,W$. If the system performs work at a rate of $200\,W$. The rate at which internal energy of the system increases $.......\,W$View Solution
- 3If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 4A polyatomic gas $\left( {\gamma = \frac{4}{3}} \right)$ is compressed to $\frac{1}{8}$ of its volume adiabatically. If its initial pressure is ${P_o}$, its new pressure will beView Solution
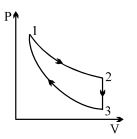
- 5Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 6$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours. - 7Under isothermal condition, the pressure of a gas is given by $P = aV ^{-3}$, where $a$ is a constant and $V$ is the volume of the gas. The bulk modulus at constant temperature is equal to $..........\,P$View Solution
- 8Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 9View SolutionThe internal energy of the gas increases In
- 10A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
