An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?
JEE MAIN 2021, Medium
$\Delta \mathrm{Q}=\Delta \mathrm{U}+\Delta \mathrm{W}$
$\frac{\Delta \mathrm{Q}}{\Delta \mathrm{t}}=\frac{\Delta \mathrm{U}}{\Delta \mathrm{t}}+\frac{\Delta \mathrm{W}}{\Delta \mathrm{t}}$
$\frac{6000}{60} \frac{\mathrm{J}}{\mathrm{sec}}=\frac{2.5 \times 10^{3}}{\Delta \mathrm{t}}+90$
$\Delta \mathrm{t}=250 \,\mathrm{sec}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
- 2View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 3View SolutionIn practice, all heat engines have efficiency less than that of a Carnot engine because
- 4In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
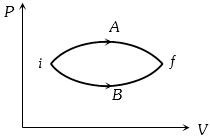
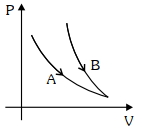
- 5Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
- 6The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
- 7Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
- 8An ideal gas expands according to the law $P^2 V=$ constant. The internal energy of the gasView Solution
- 9An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
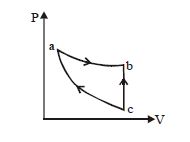
- 10A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$View Solution
