In practice, all heat engines have efficiency less than that of a Carnot engine because
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
- 2In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 3A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 4In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 5The adjoining figure shows the $P-V$ diagram for a fixed mass of an ideal gas undergoing cyclic process. $AB$ represents isothermal process and $CA$ represents isochoric process.Which of the graph shown in the following figures represents the $P-T$ diagram of the cyclic process ?View Solution
- 6Let $\eta_{1}$ is the efficiency of an engine at $T _{1}=447^{\circ}\,C$ and $T _{2}=147^{\circ}\,C$ while $\eta_{2}$ is the efficiency at $T _{1}=947^{\circ}\,C$ and $T _{2}=47^{\circ}\,C$. The ratio $\frac{\eta_{1}}{\eta_{2}}$ will be.View Solution
- 7A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
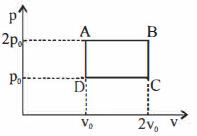
- 8A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 9A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic $($perfect thermal conductor$)$ frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top $($in $m )$ will be $. . . . . ($take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1} ).$View Solution
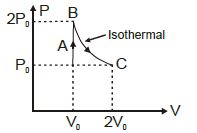
- 10The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution