A sample of gas with $\gamma=1.5$ is taken through an adiabatic process in which the volume is compressed from $1200\, {cm}^{3}$ to $300\, {cm}^{3}$. If the initial pressure is $200\, {kPa}$. The absolute value of the workdone by the gas in the process $= \,..... J.$
JEE MAIN 2021, Diffcult
$\gamma=1.5$
$p_{1} v_{1}^{\gamma}=p_{2} v_{2}^{\gamma}$
$(200)(1200)^{1.5}=P^{2}(300)^{1.5}$
$P_{2}=200[4]^{3 / 2}=1600 {kPa}$
$\mid$ W.D. $\mid=\frac{{p}_{2} {v}_{2}-{p}_{1} {v}_{1}}{v-1}=\left(\frac{480-240}{0.5}\right)=480 {J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
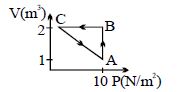
- 2An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
- 3The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
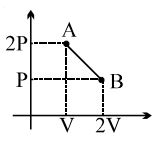
- 4In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $30$ joules of heat and $10$ joules of work was done on the gas. If the initial internal energy of the gas was $30$ joules, then the final internal energy will be ........ $J$View Solution
- 5A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
- 6View SolutionA Camot cycle consists of
- 7In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
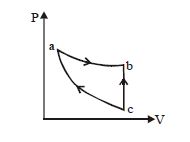
- 8An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
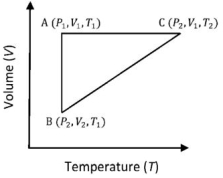
- 9A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 10View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
