An enclosed ideal gas is taken through a cycle as shown in the figure. Then
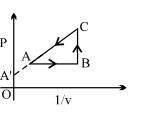

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
- 2View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
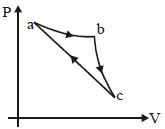
- 3In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?View Solution
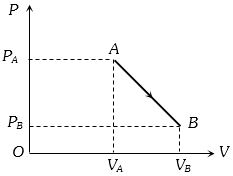
- 4An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution
- 5View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 6View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
- 7Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
- 8A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 9View SolutionWhich one is the correct option for the two different thermodynamic processes ?
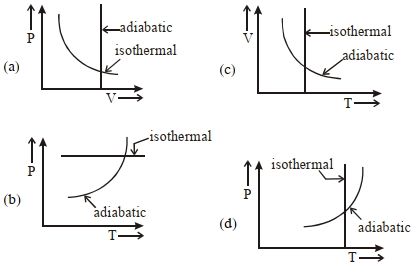
- 10A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
