Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
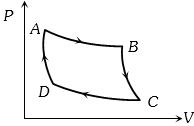
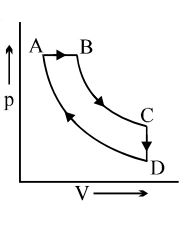
- 1Carnot cycle (reversible) of a gas represented by a Pressure-Volume curve is shown in the diagramConsider the following statementsView Solution
$I.$ Area $ABCD =$ Work done on the gas
$II.$ Area $ABCD =$ Net heat absorbed
$III.$ Change in the internal energy in cycle $= 0$
Which of these are correct
- 2$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):View Solution
- 3Unit mass of a liquid with volume ${V_1}$ is completely changed into a gas of volume ${V_2}$ at a constant external pressure $P$ and temperature $T.$ If the latent heat of evaporation for the given mass is $L,$ then the increase in the internal energy of the system isView Solution
- 4A heat engine operates between a cold reservoir at temperature ${T}_{2}=400\, {K}$ and a hot reservoir at temperature ${T}_{1} .$ It takes $300 \,{J}$ of heat from the hot reservoir and delivers $240\, {J}$ of heat to the cold reservoir in a cycle. The minimum temperature of the hot reservoir has to be $....{K}$View Solution
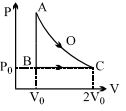
- 5An ideal gas is taken from point $A$ to point $C$ on $P-V$ diagram through two process $AOC$ and $ABC$ as shown in the figure. Process $AOC$ is isothermalView Solution
- 6View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
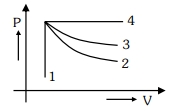
- 7An ideal gas undergoes four different processes from the same initial state as shown in the figure below. Those processes are adiabatic, isothermal, isobaric and isochoric. The curve which represents the adiabatic process among $1,2,3$ and $4$ isView Solution
- 8A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 9A vessel containing $5\, litres$ of a gas at $0.8 \,pa$ pressure is connected to an evacuated vessel of volume $3$ litres. The resultant pressure inside will be ...... $pa$ (assuming whole system to be isolated)View Solution
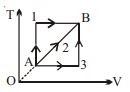
- 10A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution