An engine runs between a reservoir at temperature $200 \,K$ and a hot body which is initially at temperature of $600 \,K$. If the hot body cools down to a temperature of $400 \,K$ in the process, then the maximum amount of work that the engine can do (while working in a cycle) is (the heat capacity of the hot body is $1 \,J / K )$
KVPY 2020, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 2The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 3A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 4$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
- 5An ideal gas, initially in state $\left( P _{12}, V _1, T _1\right)$ is expanded isobarically to $\left( P _{12}, V _2, T _2\right)$, then adiabatically $\left( P _{34}, V _3, T _3\right)$. It is then contracted isobarically to $\left( P _{34}, V _4, T _4\right)$ and finally adiabatically back to the initial state. The efficiency of this cycle isView Solution
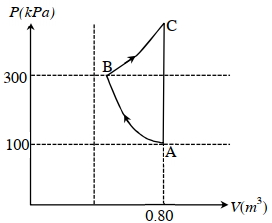
- 6In the given $P$-V diagram, a monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is first compressed adiabatically from state $A$ to state $B$. Then it expands isothermally from state $B$ to state $C$. [Given: $\left(\frac{1}{3}\right)^{0.6} \simeq 0.5, \ln 2 \simeq 0.7$ ].View Solution
Which of the following statement($s$) is(are) correct?
$(A)$ The magnitude of the total work done in the process $A \rightarrow B \rightarrow C$ is $144 kJ$.
$(B)$ The magnitude of the work done in the process $B \rightarrow C$ is $84 kJ$.
$(C)$ The magnitude of the work done in the process $A \rightarrow B$ is $60 kJ$.
$(D)$ The magnitude of the work done in the process $C \rightarrow A$ is zero.
- 7During the adiabatic expansion of $2 \,moles$ of a gas, the internal energy was found to have decreased by $100 J$. The work done by the gas in this process is ..... $J$View Solution
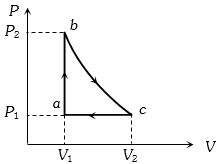
- 8Carbon monoxide is carried around a closed cycle $abc$ in which bc is an isothermal process as shown in the figure. The gas absorbs $7000 J$ of heat as its temperature increases from $300 K$ to $1000 K$ in going from $a$ to $b$. The quantity of heat rejected by the gas during the process $ca$ is ..... $J$View Solution
- 9For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
- 10During the adiabatic expansion of $2$ moles of a gas, the internal energy of the gas is found to decrease by $2$ joules, the work done during the process on the gas will be equal to ....... $J$View Solution
